A SimpleTwig Article: Galvanic Corrosion, sometimes referred to as Galvanic Action, describes the corrosion to a metal when that metal is in contact, either directly or with the help of a catalyst or another element, with another metal whose characteristics are not complimentary. Often two different metals can come in contact with one another without any corrosion, but some combinations do lead to corrosion and ultimately failure. This can be critical to prevent ‘rusting’ and unsightly stains, to prevent water penetration when a flashing fails, or to even prevent parts of a building from falling apart when a fasteners like a screws fails.
We take a look at what it is and how to prevent this type of corrosion so our buildings will last many decades longer.
The corrosion occurs when the two metals have different electrical characteristics and the presence of an electrolyte allows a conductive path to form between the two metals. This path allows for metallic ions to migrate from the anode (positively charged metal) to the cathode (negatively charged metal), thus weakening the anode and making it more susceptible to rust. Specifically, it is due to an electrochemical reaction that occurs when two dissimilar metals make contact in the presence of an electrolyte like sulfuric acid or sodium acetate.
This is why wire has a protective insulation wrap to protect its current from jumping to other metals. Think of it as understanding that all metals have conductivity potential. It’s the differences in electrode potential that drives the corrosive attack on the positively charged metal (anode), forcing it to dissolve into the electrolyte. This action is much the same way heat will transfer to colder side of a wall, humans using the resistance of insulation to slow down this transfer, we noting the quality of this resistance as R factors. In this way, even two ‘bad’ metals can be close to one another if there is an insulator preventing the transfer of electricity.
Still, as noted, some different metals can touch one another without issue. In construction it is imperative to know what metals can rest next to one another, and which combinations must either be insulated or placed far enough apart to prevent corrosion.
Ironically in concrete where sodium acetate that’s used to seal concrete and protect it from weathering can cause galvanic corrosion in metals installed in or around it. In this case the sodium acetate isn’t acting as a resistor (or insulation) but as a catalyst allowing the corrosive action to accelerate.
In this way it is important to understand that it is not just unlike metals that touch, but that these metals ‘touch’ or are close enough to have ions flow between them through a ‘solution’. The solution can be acid (as in acid rain), a saline (as in salt water) or an alkaline solution, and as such essentially recreates the action of a battery, that is the flow of electrons between the two metals.
Rain, Water, Humidity, the Third Partner:
Rain ranges from being slightly acidic to very much so, especially in larger cities where vehicles and trucking give off pollution. Putting it in perspective, acidic rain makes what it touches less alkaline in terms of Ph levels. Since acidity and alkalinity are two sides of the same coin their levels waif between the two. In terms of galvanic action, either acid or alkaline rain can increase the flow of electricity between two dissimilar metals. In India, Australia and a few other parts of the world rain tends to be more alkaline.
It is important to understand that it is the electrolyte, or as defined “a liquid or gel that contains ions and can be decomposed by electrolysis” is the third element needed along with two dissimilar metals, to allow a reaction. Since most buildings get rained on, this is the contributing factor. Still, one must not dismiss the effects of humidity on the interior of a home or building, especially when one uses metal studs, dropped ceilings, metal finishes and so on to fit out a space.
BEST METHODS TO PREVENT GALVANIC CORROSION between DISSIMILAR METALS:
- Separate metals electrically by using non-conductive materials which stops ion migration:
- Separate metals with an insulator like plastic;
- Use water-repellent compounds like grease to prevent contact with electrolytes;
- Consider electroplating using noble metals* (like gold, silver or platinum) that resist corrosion better.
- Apply cathodic protection; This is the technique used to control the corrosion of a metal surface by making it the cathode of an electrochemical cell. A simple method of protection connects the metal to be protected (anode) to a more easily corroded ‘sacrificial metal’ to act as the anode. This is accomplished by physically connecting the anode that needs protecting by a wire to the sacrificial metal.
- For Aluminum and Copper, apply antioxidant paste. Antioxidant paste can easily be found on the internet. It is sometimes referred to as ‘connection grease.’
CHOOSE SIMILAR METALS to AVOID GALVANIC ACTION ALL TOGETHER:
By simply choosing metals that avoid galvanic action there will never be an issue. Yet in today’s construction world it can be easy for a contractor to mix the wrong type of fastener with the wrong metal.
In this respect, one should understand how to read the following chart which lists all metals. SimpleTwig will then try to extract the most commonly used construction metals for a separate chart, to help ensure the prevention of Galvanic Action on our projects and for those who use this article on their projects.
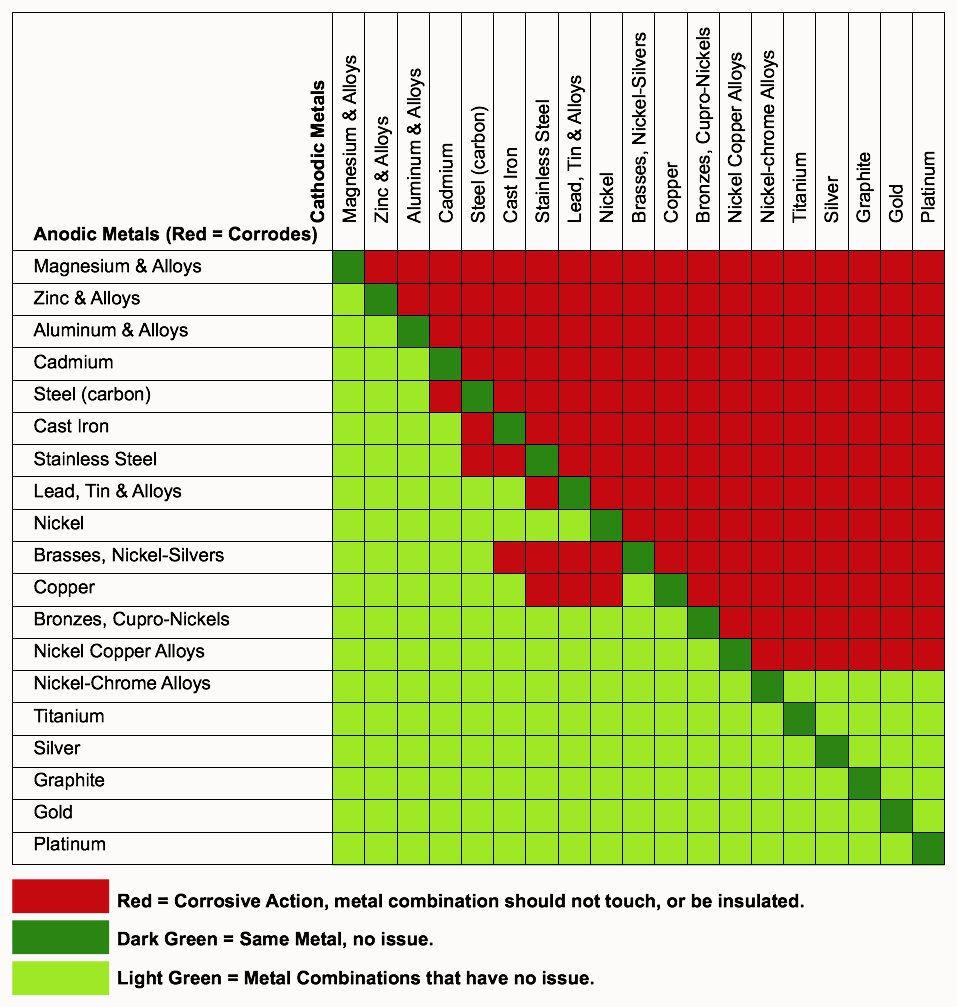
Looking at copper, often used in metal flashing, gutters and downspouts, the chart shows there’s no issue when in contact with aluminum (but see notes on copper below), steel or cast iron, but there is a problem when in contact with stainless steel, another common flashing metal.
To understand this chart, the ‘more noble’ metals are the anodic metals at the bottom of the chart, that is, while gold is a noble metal, bronze is more noble than cast iron. The least noble metal is magnesium and its alloys.
Put in another form as a list, we show the most Anodic (corrosive susceptible metals) down to the most cathodic metals (passive in that they do not corrode, but allow ions to pass if in contact with a ‘bad’ metal choice).
ANODIC – active
-
- Magnesium alloys
- Zinc
- Beryllium
- Aluminum 1100, 3003, 3004, 5052, 6053
- Galvanized steel
- Cadmium
- Aluminum 2017, 2024, 2117
- Mild Steel (1018), Wrought Iron
- Cast iron, Low alloy high strength steel
- Chrome iron (active)
- Stainless steel, 430 series (active)
- Stainless steel 302, 303, 304, 321, 347, 410, 416, (active)
- Nickel (resist)
- Stainless steel 316, 317, (active)
- Carpenter 20 CB-3 stainless (active)
- Aluminum Bronze (CA 687)
- Hastelloy C (active), Inconel 625 (active), titanium (active)
- Lead-tin solders
- Lead
- Tin
- Inconel 600 (active)
- Nickel (active)
- Brasses (naval, yellow, red, admiralty)
- Copper (CA102)
- Manganese bronze, tin bronze
- Silicon bronze
- Nickel silver
- Copper-nickel alloy
- 430 stainless steel
- Nickel (passive), aluminum, bronze
- Monel 400, K500
- Silver solder
- Nickel (passive)
- Chrome iron (passive)
- 302, 303, 304, 321, 347, stainless steel (passive)
- 316, 317, stainless steel (passive)
- Carpenter 20 CB-3 stainless (passive), Incoloy 825
- Nickel-molybdeum-chromium-iron alloy (passive)
- Silver
- Titanium and titanium alloys
- Graphite
- Zirconium
- Gold
- Platinum
Cathodic – passive
HOW CERTAIN METALS ARE USED IN CONSTRUCTION:
Cadmium:
Cadmium is often a sacrificial metal used as a coating on other metals, to prevent the main metal from corrosion. Electroplated cadmium is a robust and versatile metallic coating, a soft white metal that, when plated onto steel, cast iron, malleable iron, copper and powdered metal, functions as a protective layer to the substrate material. To enhance the corrosion protection when using cadmium plating, chromate conversion coatings can be applied over the plated metal, yielding the familiar gold color, or olive drab color.
This coating is good with bonding adhesives (aircraft industry) and for salt-water environments. More information can be found here: http://www.chemprocessing.com/page.asp?pageid=54&
Copper:
Used in flashing, gutters, downspouts and fasteners, as well as in wiring or busbars.
Aluminum is very susceptible to galvanic corrosion in contact with copper, with an electrolyte like water with some ionic content. One can tin plate the copper bolts, for instance, to help stop or slow the galvanic attack. This corrosion can be critical in airplane, but can be superficial in architectural projects in that the aluminum will corrode but that, for instance rain water isn’t highly conductive, noting that in highly polluted cities this may be a changing factor.
Fasteners:
Because bolts, screws and welds are surrounded by the materials they fasten, they can take on the role of anode risking rapid corrosion and thus should be avoided. Zine-coated fasteners use only with steel that’s coated with aluminum, zinc and galvalume. Zinc or aluminum coated fasteners should not attach copper or stainless-steel panels.
To minimize the risk, match the surface metal on both the fastener and the metal is is connected to. If necessary, match a ‘large anode’ (the metal that will rust) with a small cathode to limit corrosion, i.e. use fasteners like bolts and screws of a metal less likely to corrode or more cathodic than the metal it is attaching to.

Minimizing Galvanic Corrosion:
- use similar metals;
- prevent dissimilar metals from becoming wet, electrically connect by water, or humidity;
- keep ‘small anodes’ from touching ‘large cathodes;’
- add protective coatings, known as a sacrificial coating, to protect the primary metal and its function;
- add other coatings, like paint, plastic or other non-metallic barrier. Note that paint coatings can be scratched and thus expose the metal and make is susceptible to galvanic action if in contact with another metal.
Wood Preservatives (Decks): containing copper, mercury, or fluorides.
Do not allow aluminum, aluminum-coated, and galvalume-coated panels and trim to come into direct contact with wood preservatives which contain coper, mercury, or fluorides. Avoid direct contact between bare metal panels and treated lumber where condensation will frequently form on the metal surface in contact with lumber, and where wood treatment is ‘more noble’ than the metal surface. Use an appropriate barrier to separate the two.
Passivation:
A process, through surface cleaning and sealing, this process lowers the metal’s electrical potential and improves its corrosion behavior.
Example:
Attaching a Brass Plate with an Aluminum Plate via a 304 Stainless Steel Bolt. Both the brass and the aluminum will corrode severely where they touch the stainless steel because they are much more anodic than stainless steel. The aluminum will corrode more because it is more anodic than brass, and will also corrode where it contact the brass since brass is more cathodic.
In this case there should be a barrier between the brass and aluminum, with the selection of a bolt that is compatible to both materials.
CONCLUSION:
It is imperative on large projects that there be a professional specification writer assigned to the project, one that throughly understands what materials are being used/proposed and can make recommendations for changes, or to specify proper coatings, separations, and insulators to ensure Galvanic Action does not occur.
In smaller residential projects, often reliance on the proper use of materials falls on the Contractor, and sometimes if an Architect is involved in both shoulders to prevent metal corrosion. In either case, one should look at dominate materials of the house including a wood deck (and stainless steel screws or coated bolts), copper gutters and downspouts (using copper fasteners), aluminum thresholds, roofing fasteners, etc. to ensure these components hold up over time and function as intended structurally. In this way, staining, unsightly pitting or oxidation, or even structural failure can be prevented.
Be sure to read more of our Practical and Technology articles on our SimpleTwig Architect’s Blog. Or simply choose a new category from the list in the righthand side column. Comments are welcome and appreciated.
FOOTNOTES:
*Noble Metals include: Ruthenium, rhodium, palladium, silver, osmium, iridium, platinum, gold. These metals can act as an insulator to prevent galvanic action and thus prevent corrosion.
SOURCES:
http://www.monarchmetal.com/blog/galvanic-corrosion-common-questions-answered/
https://www.fastenal.com/content/feds/pdf/Article%20-%20Corrosion.pdf
http://www.monarchmetal.com/blog/galvanic-corrosion-common-questions-answered/
http://sciencing.com/rain-increase-alkalinity-22396.html

At the risk of sounding stupid: The chart by Chart created by SimpleTwig Architecture-2017 lists each metal on the top row & left side column.
So with the aluminium on the left, & zinc on the top, there is a red square saying there will be galvanic corrosion. However with the zinc on the left, & aluminium on the top, there isn’t galvanic corrosion.
So if I have a copper pipe connected to an aluminium reservoir, both being in contact with the water stream, will there, or will there not be galvanic corrosion?
The column on the left are Anodic Metals while those along the top are Cathodic Metals. Anodic generally means the surface will create an oxide finish, like an anodized surface. Cathodic is generally a metal that controls the corrosion of a metal surface by making it the cathode of an electrochemical cell. I would suggest looking at wikipedia for more detailed information. Regarding copper and aluminum the chart says there will be Corrosive Action. The above is a general reference.
How we could do to prevention corrosion in daily life?
One of the best to prevent metals and metallic surface from corroding is to apply aluminized coatings. They have strong protection effect against corrosion.
Thanks Alex for the input. I see this is the service you provide: http://www.vaporkote.com/ Good luck.